We often receive questions from ketamine clinics regarding who must obtain a DEA registration to dispense controlled substances. There are two candidates to consider – the healthcare providers who work at the clinic and the clinic itself. Often, there is a misconception about whether a clinic needs to hold a DEA registration. We wrote about this briefly previously and also about a specific DEA registration exception. But today we discuss the DEA registration requirements in more detail.
Who Needs to Hold a DEA Registration?
The general rule for who must hold a DEA registration is set forth in the federal Code of Federal Regulations (CFR), which provides as follows:
Every person who manufactures, distributes, dispenses, imports, or exports any controlled substance or who proposes to engage in the manufacture, distribution, dispensing, importation or exportation of any controlled substance shall obtain a registration unless exempted by law or pursuant to [21 CFR] §§ 1301.22 through 1301.26. Except as provided in paragraph (b) of this section, only persons actually engaged in such activities are required to obtain a registration; related or affiliated persons who are not engaged in such activities are not required to be registered. (For example, a stockholder or parent corporation of a corporation manufacturing controlled substances is not required to obtain a registration.)” 21 CFR 1301.11(a).
The operative question is who is manufacturing, distributing, dispensing, importing, or exporting ketamine for a ketamine clinic? This can include a lot of people! There are certain components that can be quickly eliminated for most ketamine clinics, including manufacturing, importing, or exporting ketamine. Those functions are typically left to other entities, and not the ketamine clinic nor the healthcare providers who prescribe and dispense the actual ketamine.
However, like most things under the law, answers are not always so obvious. Thus, a review of certain definitions helps to analyze these issues. The important definitions include:
- “Dispense” which means delivering “a controlled substance to ultimate user.” 21 USC 802(10).
- In turn, an “ultimate user” “means a person who has lawfully obtained, and who possesses, a controlled substance for his own use or for the use of a member of his household….” 21 USC 802(27)).
- A “dispenser” is “an individual practitioner, institutional practitioner, pharmacy or pharmacist who dispenses a controlled substance.” 21 C.F.R. § 1300.01(b)(11).
- An “individual practitioner” is defined as “a physician, dentist, veterinarian, or other individual licensed, registered, or otherwise permitted, by the United States or the jurisdiction in which he/she practices, to dispense a controlled substance in the course of professional practice, but does not include a pharmacist, a pharmacy, or an institutional practitioner.” 21 C.F.R. § 1300.01(b)(17).
- An “institutional practitioner” is defined as a licensed or registered hospital. 21 C.F.R. § 1300.01(b)(18).
Applying the foregoing to healthcare providers demonstrates why such providers need to hold a DEA registration. A healthcare provider “dispenses” ketamine to the “ultimate user” which is the patient. Moreover, a review of the other definitions set forth above is often necessary.
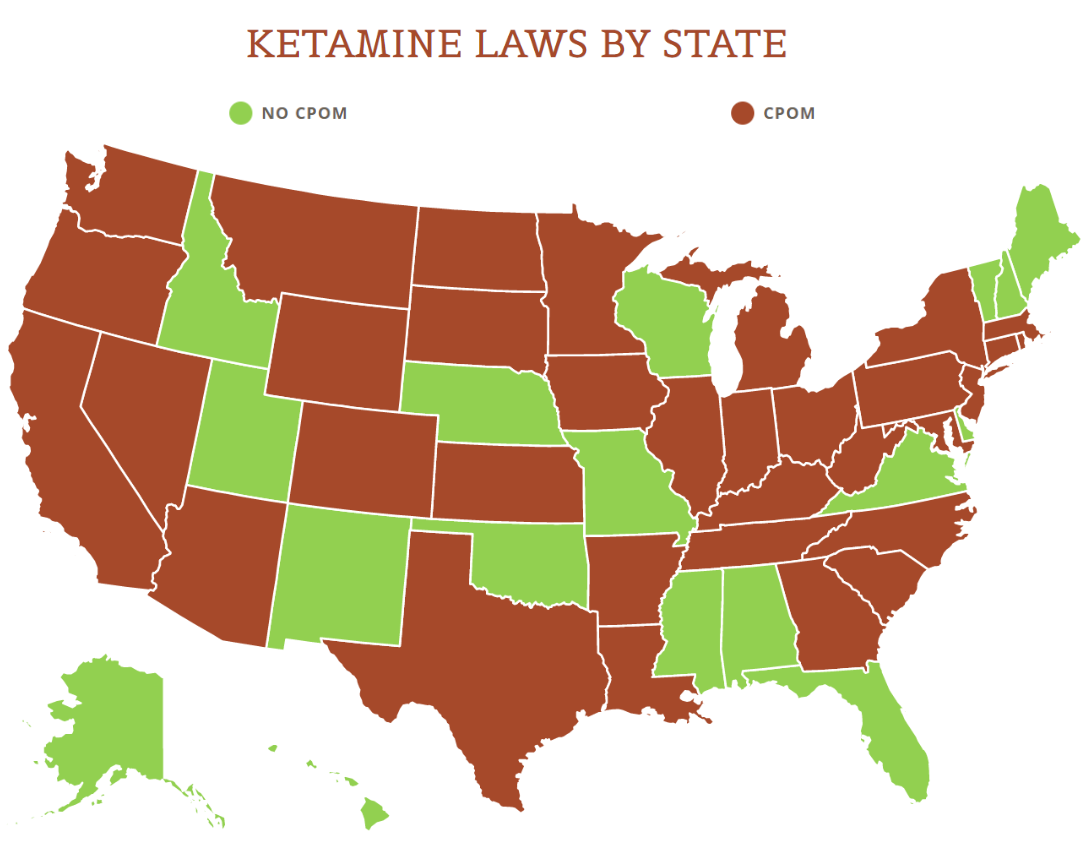
View the US Map of Ketamine Legality
Do Ketamine Clinics Need to Hold a DEA Registration?
Unlike healthcare providers, most clinics do not need separate DEA registrations. Instead, such clinics can rely upon their healthcare provider’s DEA registration (if, among other things, the healthcare providers register the location of each clinic or office with the DEA).
The reasons why a ketamine clinic does not need to hold a DEA registration require the same analysis as set forth above. A ketamine clinic does not “dispense” ketamine to the “ultimate user” – only a provider can dispense drugs to a patient. While the clinic and its employees can order controlled substances and assist with ensuring the healthcare provider complies with all DEA laws and regulations, the clinic cannot then dispense the drug to the patient.
While the ketamine clinic may be able to register with the DEA, often, it is not necessary to do so.
The InterDent Case and DEA Registrations
We have only been able to find one case that helps to define these requirements, Kim v. Interdent, Inc., 2010 WL 3155011 (N.D. Ca. 2010) (the “InterDent” case). InterDent was a managed services organization (MSO) that worked with professional corporations (PC) owned by dentists. In the InterDent decision, “Plaintiff alleges that InterDent was required to be registered under the [Controlled Substances Act], that InterDent failed to provide effective controls and procedures, and that Dr. Bae was a member of the class of people whom the regulations were designed to protect.” The court in InterDent went through the same analysis that is set forth above to determine that InterDent did not need to be registered with the DEA.
Another issue raised in the InterDent decision was whether InterDent was involved in the distribution of a controlled substance. The court noted:
The [Controlled Substances Act] states “‘distribute’ means to deliver (other than by administering or dispensing) a controlled substance or a listed chemical. The term ‘distributor’ means a person who so delivers a controlled substance or a listed chemical.” 21 U.S.C. § 802(11). “Deliver,” in turn, is defined as “the actual, constructive, or attempted transfer of a controlled substance or a listed chemical, whether or not there exists an agency relationship.” 21 U.S.C. § 802(8). Here, InterDent employees placed orders and paid for controlled substances on behalf of Dr. Bae and other registered dentists, for use in the dental practice. However, the registration requirement is waived “for any agent or employee of a person who is registered to engage in any group of independent activities, if such agent or employee is acting in the usual course of his/her business or employment.” 21 C.F.R. § 1301.22(a). Under the dental group management agreement between InterDent and the PCs, InterDent “serve[s] as the purchasing agent for [PCs]” and does not possess dental licenses or certificates. [….] Thus, pursuant to the terms of the management agreements and 20 C.F.R. § 1301.22(a), InterDent is exempt from the registration requirement because its employee acted as purchasing agents for the dentists.
The last portion of the foregoing quote is an important lesson for drafting management services agreements (MSA). The court obviously found that the MSA between InterDent and the PCs was very material. If your ketamine clinic uses an MSO, it would be prudent to ensure the MSA includes language that reflects the parties’ responsibilities when it comes to ordering ketamine. The operative language for the MSA should include that the MSO “serve[s] as the purchasing agent for [PCs]”.
These are not easy issues. An analysis of the MSO/PC relationship and responsibilities will dictate who needs to hold a DEA registration for ketamine. A thorough review of the MSA, the underlying facts, and the law should provide the answers regarding who needs to hold a DEA registration.